Glaucoma is a progressive blinding disease characterized by loss of retinal ganglion cells (RGCs), neurons that transmit visual information from the eye to the brain. All existing glaucoma treatments focus on lowering the intraocular pressure, which is a risk factor for glaucoma. However, there are no clinically used therapies that directly promote RGC survival. Our research is focused on understanding the role of neuroinflammation in pathogenesis of glaucoma, with the goal of developing new therapies for this devastating blinding disease.
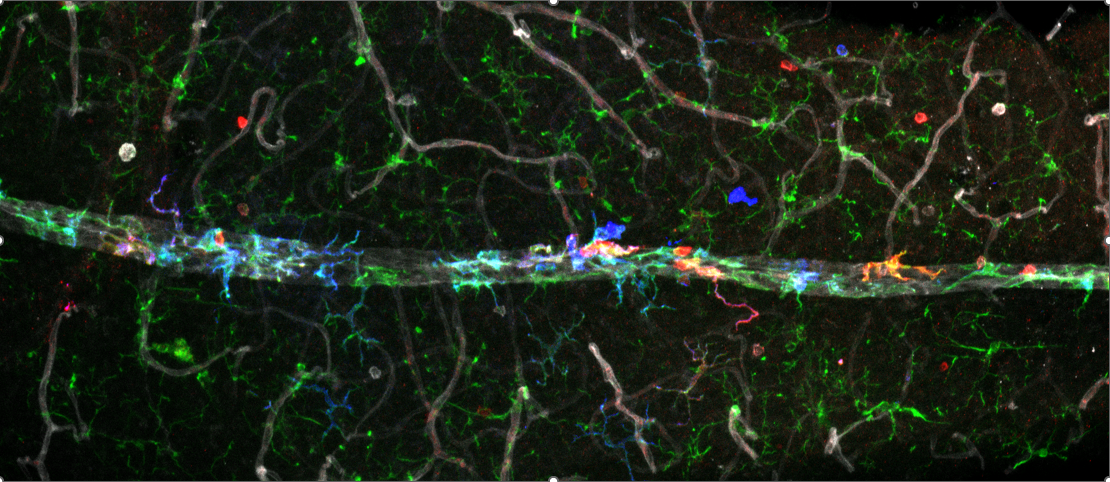
In our previous work, we discovered that microglia, the resident immune cells of the central nervous system, switch from a homeostatic to a neurodegenerative molecular phenotype in glaucoma that is shared with neurodegenerative diseases of the brain. One of the genes upregulated in microglia in glaucoma is Apoe, the major lipoprotein in the brain. Notably, the APOE4 allele increases the risk of Alzheimer’s disease but decreases the risk of glaucoma. In the manuscript we published in Immunity, we demonstrate that Apoe is the key upstream regulator of the microglial response in glaucoma, while a secreted inflammatory molecule Galectin-3 is an important effector that can be targeted genetically and pharmacologically to prevent RGC loss in glaucoma.
Furthermore, APOE and Galectin-3 are upregulated in the retina and aqueous humor of glaucoma patients. We also demonstrate that, similarly to Apoe loss-of function, mice with the APOE4 allele have impaired microglial activation and improved RGC survival despite elevated intraocular pressure, explaining why this allele is associated with a decreased risk of human glaucoma. In addition to its relevance to glaucoma, this research also has interesting implications for the pathophysiology of Alzheimer’s disease, resulting in recent publications we co-authored in Nature Neuroscience and Nature Immunology. As a result of these efforts, our work has been profiled by Mass General Brigham in the New York Times as an example of collaboration and innovation across different MGB institutions.
Our current research is focused on using cutting-edge technologies such as single cell RNA sequencing and lineage tracing to further elucidate the immune cell landscape in the retina and the optic nerve in health and disease. We continue our translational mission by developing new therapeutic approaches for targeting neuroinflammation in glaucoma, as well as investigating potential biomarkers of glaucoma in the aqueous humor and serum of glaucoma patients. Finally, through a collaboration with several investigators at MGH, we are developing a novel eyedrop-based diagnostic for Alzheimer’s disease.